The mechanical behavior of materials is crucial for both safety and economic aspects. Since the production of engineering products are made via utilizing appropriate grade of materials, the mechanical approval of the used materials must be done before the assembly.
When the design of the engineering product is conducted, the type of the used material becomes a significant concern for the engineers. The fracture progress of the materials (mostly metals for general purposes) must be slowed to prevent sudden failures. At this point, a reminder for the fracture types may be advantageous for further examinations on ductile-brittle transition temperature. Fracture types of the metals can be classified as ductile and brittle fracture, which strongly differs from the other in the manners of propagation and energy capacities. Ductile fracture mostly consumes a high amount of energy during the progress of fracture.
Since the toughness ability of the ductile materials is higher than the brittle ones, the required energy for the failure of ductile materials is usually more than the necessary energy for the brittle materials. Therefore, the failure propagation occurs faster for the brittle materials than the ductile materials. Moreover, during the deformation of the ductile materials, one can be observed a substantial amount of plastic deformation. The plastic deformation of the ductile metals is crucial since the shape change of the metal can be considered as an alert for the possible failure of the metal. In contrast with that, plastic deformation of brittle metals can be barely observed since the sudden brittle fracture of the metals comes with a low energy consumption during failure. Therefore, for most of the applications, the ductile type of fractures is desired for the detection and control of the possible failures.
Furthermore, the point of temperature where the material’s behavior changes from ductile to brittle or vice versa is vital for designing the products. The temperature point where the material experiences the change from either ductile to brittle or brittle to ductile is called as ‘Ductile to Brittle Transition Temperature’ (DBTT). Information about the ductile-brittle transition temperature of a certain metal (material) may prevent possible disasters. A solid example of the importance of the ductile-brittle transition temperature can be given as the failure of the Liberty ship during World War II. The transition temperature of the metal of the ship was miscalculated during the production. When the ship sailed in the cold sea-waters, the metal body of the ship suddenly changed from ductile to brittle. The flaws near the weld seams of the ships acted as stress-concentrated points. Since the ductile-brittle transition temperature of the steel grade miscalculated, a sudden ductile to brittle transformation emerged, and steel became brittle. Moreover, the crack propagation rate excessively increased because of the brittle transformation of the steel body of the ship. Finally, a sudden fracture of the ship occurred. The below figure shows the Liberty ship disaster.

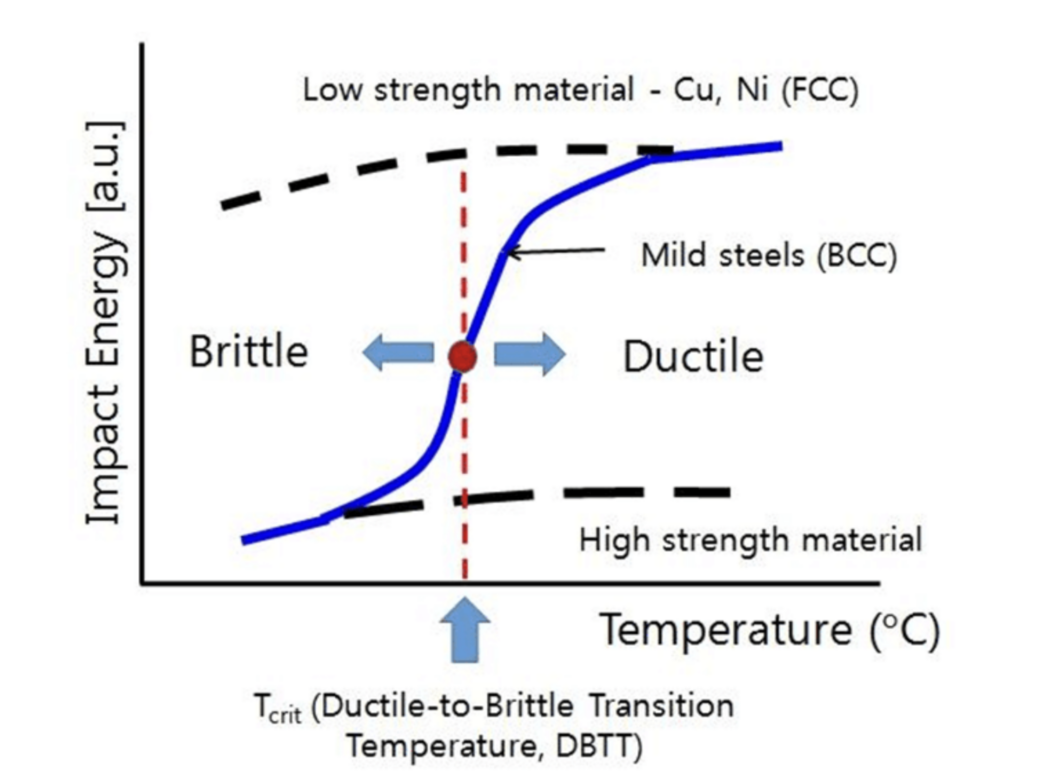
For most engineering applications, one must prefer a low ductile-brittle transition temperature to ensure a wider ductility range. Utilization of the wider ductility range is desired to inhibit the sudden cracks and failures in the metal body. In addition to that, the crystal structure of the selected material (metal) is also crucial in the manner of ductile-brittle transition. The ductile-brittle transition of FCC metals is smooth when compared with the sharp transition curve of the BCC metals. The reason for this phenomenon can be explained by considering the Peierls stress-temperature relationship for FCC and BCC materials. A brief reminder of Peierls stress must be given before understanding the temperature dependence of Peierls stress. High Peierls stress in given materials results in difficult plastic deformation. When the temperature of the environment decreases, Peierls stresses for both FCC and BCC materials increase. However, the increase in the Peierls stress for BCC materials is immensely greater when compared with the increase of Peierls stress for FCC materials. Hence, greater Peierls stress leads to brittle fracture of the materials. The sharp transition of the ductile-brittle transition fo BCC materials can be explained by taking the Peierls stress- temperature relationship into consideration. The change of the ductile-brittle transition for BCC and FCC materials can be seen below figure.
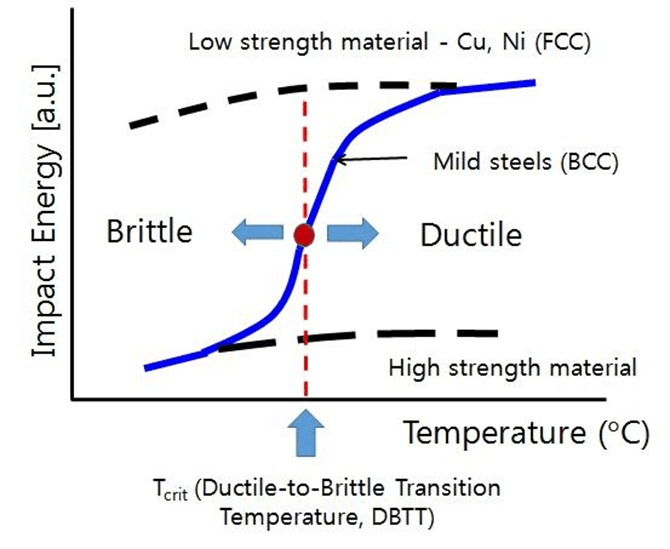
The desired properties of used metals in particular applications must be optimized during the production step. One of the essential features of the metals can be stated as toughness. In addition to energy absorption ability (toughness), lower ductile-brittle transition temperature must be ensured to widen the ductile range of the metal. For steel materials, reducing the carbon amount and refining the grain size may contribute to the decrement of the ductile-brittle transition temperature. Moreover, the homogenous microstructure of steel can decrease the transition temperature. The reduction of carbon amount and grain refinement also increases the toughness of the steel grades. Furthermore, inclusions in the matrix of the steel must be decreased to enhance the toughness property.
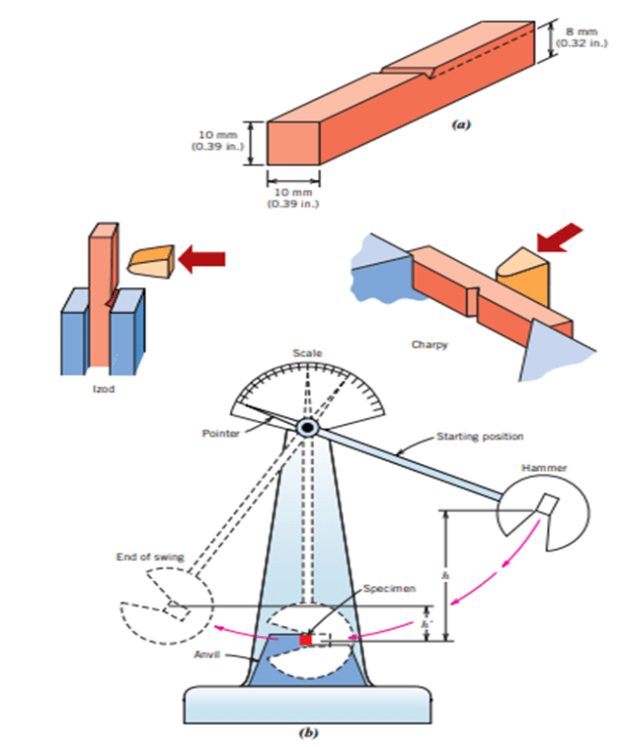
To determine the ductile-brittle transition temperature of specific metals, impact testing methods are used. Two tests are commonly used for the detection of the transition temperature and impact energy absorption ability, which is Charpy and Izod methods. In the Charpy V-Notch test, a specimen is placed vertically at the base of the test device. A V-shaped notch is machined on the sample to provide the stress concentration and fracture of the material. The fundamental theory of both Charpy and Izod tests are based on the change of potential energy amount. At the initial stage of the Charpy test, a free-falling pendulum with a certain mass (m) is placed at a certain height (h). When the pendulum strikes through the test specimen, some potential energy is absorbed by the specimen for the fracture. It is evident that the height of the pendulum after the collision will be lower than its initial position. The potential energy change after the strike of the pendulum can be determined by considering the initial and final height of the pendulum. Thus the difference in the potential energy is assumed as the impact energy absorption ability or toughness of the specimen. The determination of the ductile to brittle transition temperature is done by conducting an impact-energy test under different temperatures. The fracture of the material is observed at various temperatures and is decided whether it is ductile or brittle. The change of the fracture behavior from brittle to ductile is examined when the absorbed energy dramatically decreased. The resulting change range of the temperature is considered as the ductile to the brittle transition temperature. The second common testing method is the Izod test. The most significant difference between the Izod test from the Charpy test is the placement of the specimen at the base of the test device. In contrast with the Charpy test, in the Izod test, the specimen is placed vertically to the bottom. The Izod test method is mostly used for testing the impact energy of polymeric materials. The below figure shows the impact testing apparatus and specimen.
Read more: Brittle and Ductile Fracture
Bibliography
- Ductile to brittle transitions in materials. (2020). Retrieved November 2020, from https://www.jeremyjordan.me/ductile-to-brittle-transitions-in-materials/
- Callister, W. (2013). Materials Science and Engineering (9th ed., pp. 253, 271). New York: Wiley-Blackwell.
- Chao, Y. J., Ward, J. D., & Sands, R. G. (2007). Charpy impact energy, fracture toughness and ductile–brittle transition temperature of dual-phase 590 Steel. Materials & Design, 28(2), 551–557. doi:10.1016/j.matdes.2005.08.009
- Tanguy, B., Besson, J., Piques, R., & Pineau, A. (2005). Ductile to brittle transition of an A508 steel characterized by Charpy impact test. Engineering Fracture Mechanics, 72(1), 49–72. doi:10.1016/j.engfracmech.2004.03.010